- Blog
- Sims 4 traits mod pack
- Pokemon egglocke download gba
- Technical brutal death metal font
- Autocad 2005 works on windows 10 but not 2008
- Gatchaman 2013 subtitle track
- Friends series finale date
- Ultraman cosmos powers
- Gaussian 09w e01
- Global entry login issue
- Sampletank 3 no instrumsnts
- Pavani reddy first husband
- Topaz denoise 6 camera preset profile pentax
- A form b form z form dna
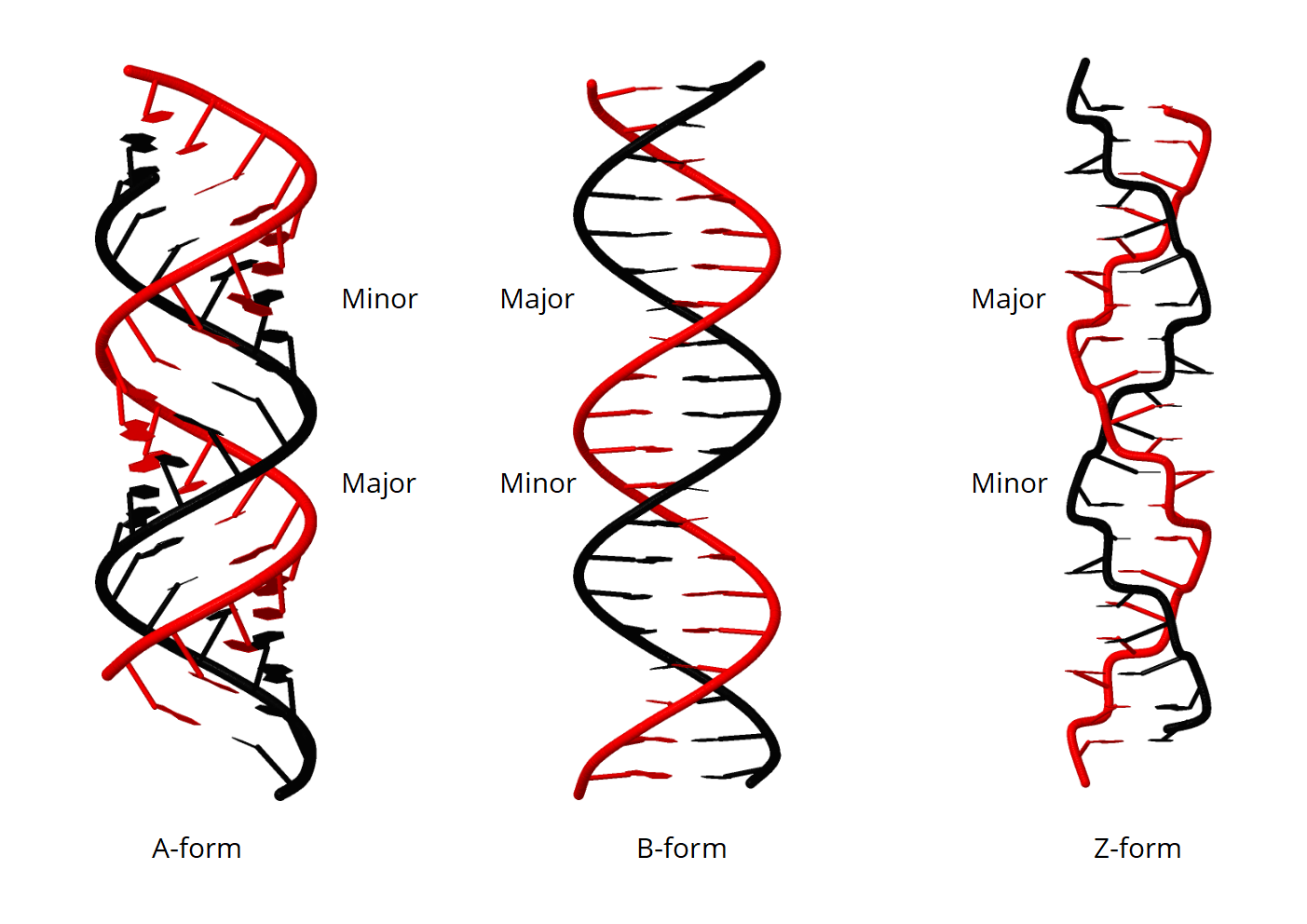
The explanation for this effect was provided by the subsequent determination of the corresponding three-dimensional structure through x-ray diffraction ( 1, 8), resulting in a left-handed helicity with an alternating anti (pyrimidine) and syn (purine) orientation of the nucleotide bases together with Watson-Crick basepairing ( 1). However, already in 1972, Pohl and Jovin ( 7) were able to observe an almost complete inversion of the ultraviolet circular dichroism (CD) spectrum of poly(GC) double-helical oligonucleotides upon addition of large amounts of salt (e.g., 4 m NaCl). The A-DNA conformation is the one favored under low hydration or moderately high ionic-strength conditions, while the B-DNA conformation is preferred otherwise. Under physiological (low-salt) conditions, DNA preferentially adopts B or A conformations (along with ∼20 sequence-dependent variations of these two forms ( 3– 5)), characterized by Watson-Crick basepairing, anti orientation of the nucleotide bases, and a right-handed helicity ( 3, 6). Yet, the conformational pathway(s) followed during the interconversion between B- and Z-DNA remain experimentally unknown at the atomic level. As a result of this research, the occurrence of Z-DNA in vivo is now widely recognized, and the major thermodynamic factors influencing its stability are well understood.
+Comparison+of+A%2C+B%2C+and+Z+forms+of+DNA.jpg)
Since the accidental discovery of the left-handed Z-DNA form in the late 1970s ( 1), numerous experimental studies have investigated the relative stability of this DNA form, the influence of environmental factors on this stability, and the possible role of Z-DNA conformations in biological processes ( 2).

The transition between the B and Z conformations of double-helical deoxyribonucleic acid (DNA) belongs to the most complex and elusive conformational changes occurring in biomolecules. The results suggest that for this oligomer length and sequence, the transition mechanism involves: 1), a stretched intermediate conformation, which provides a simple solution to the important sterical constraints involved in this transition 2), the transient disruption of Watson-Crick hydrogen-bond pairing, partly compensated energetically by an increase in the number of solute-solvent hydrogen bonds and 3), an asynchronous flipping of the bases compatible with a zipperlike progression mechanism. In this article, we report simulations of this transition for the 3′-(CGCGCG)-5′ hexamer duplex using targeted molecular dynamics with the GROMOS96 force field in explicit water under different ionic-strength conditions. While the occurrence of Z-DNA in vivo is now widely recognized and the major factors influencing its thermodynamical stability are largely understood, the intricate conformational changes that take place during the B-to-Z transition are still unknown at the atomic level. Since the accidental discovery of the left-handed Z-DNA form in the late 1970s, research on this DNA morphology has been engaged in resolving questions relative to its stability, occurrence, and function in biological processes.
